Clinical Trials
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

China Biotech Podcast: AACR, BIOSECURE European Perspectives
Guest Andrew McConaghie from Scrip's UK team joins Brian Yang and Dexter Yan to discuss AACR highlights from Chinese biotechs, European perspectives on the proposed US BIOSECURE Act, two recent acquisitions and Boehringer Ingelheim's recent quarterly results.

AbbVie Claims Rinvoq Superiority To Dupixent In AD
In a head-to-head study of Rinvoq versus Dupixent in atopic dermatitis, the AbbVie drug showed superiority on a composite endpoint measuring skin clearance and itch resolution.

Falling Sales Force New Priorities At Roche
The Swiss major brings a new ruthlessness to the compounds in its pipeline: if they’re not first or best, they’re out.

IGI Bets On Trispecific Antibody To Make Inroads Into Big Pharma Myeloma Turf
Ichnos Glenmark Innovation’s president and CEO talks to Scrip about the promising activity profile of the alliance’s early stage trispecific antibody versus Janssen’s teclistamab and also maintains that the setback for Gilead’s magrolimab hasn’t eclipsed prospects for its bispecific antibody.

Chinese Firms Up Their Game In Novel Flu Antiviral Development
Joincare Pharmaceutical and partner TaiGen Biotechnology tout preliminary Phase III results in uncomplicated acute influenza for TG-1000, a homegrown follower of Shionogi/Roche’s oral antiviral Xofluza. Novel antivirals for flu were hotly pursued by Chinese developers throughout 2023.

Sanofi Clinches Key Phase III Immunology Win With Rilzabrutinib
Sanofi’s BTK inhibitor – one of two from its 2021 Principia buy – passed a Phase III test in immune thrombocytopenia as the company focuses on Dupixent-like blockbusters going forward.

BMS Has A Strong Pipeline, But Access Challenges Remain
Bristol Myers Squibb’s head of major markets, Monica Shaw, wants to improve patients’ access to lifesaving therapies. And the group has several new products ready to roll.

Corbus And Janux Lead Q1’s Small-Cap Winners
Corbus has presence in both antibody drug conjugates and obesity, and was the stand-out small-cap target for investors with a fivefold increase in its share price in Q1.

Quick Listen: Scrip's Five Must-Know Things
In this week's podcast edition of Five Must-Know Things: Teva’s immunology biosimilar launches; Intra-Cellular’s promising depression data; Phase III sleep apnea win for Lilly’s tirzepatide; an interview with Viking’s CEO; and China looks to define innovative drugs.

Sanofi Skates To Where The Puck Is With Immunology
The French drugmaker has not abandoned oncology but has made it very clear that immunology and inflammation is the priority for its R&D euros.

Pipeline Watch: Five Approvals And 23 Phase III Updates
Pipeline Watch is a weekly snapshot of selected late-stage clinical trial events and approvals announced by pharmaceutical and biotech companies at medical and industry conferences, in financial and company presentations, and in company releases and statements.

Parkinson’s Results Further Sweeten AbbVie’s Acquisition of Cerevel
Cerevel, which AbbVie is spending $8.7bn to acquire, unveiled topline results from the TEMPO-3 study of tavapadon, which an analyst said represents potentially a $1bn opportunity.
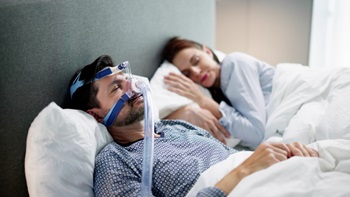
Lilly Can Rest Easy As Tirzepatide Scores Phase III Sleep Apnea Win
Topline results from two studies in obstructive sleep apnea among obese adults showed efficacy crossing the 50% threshold that physicians have called clinically meaningful.

Sage’s Dalzanemdor Prospects In Doubt After First Mid-Stage Readout
Dalzanemdor (SAGE-718) showed no difference from placebo in a Phase II Parkinson’s disease study, raising concerns about readouts later this year in Huntington’s and Alzheimer’s diseases.

MRM Health Pulls A Pouchitis Hit Out Of The Bag
The mid-stage success is encouraging, but taking on Takeda won’t be easy.

First Phase III Depression Data For Intra-Cellular’s Caplyta Exceed Expectations
With positive results from the first Phase III trial of lumateperone in major depressive disorder and a second readout expected later this quarter, the company plans a supplemental filing in late 2024.
You must sign in to use this functionality
Authentication.SignIn.HeadSignInHeader
Email Article
All set! This article has been sent to my@email.address.
All fields are required. For multiple recipients, separate email addresses with a semicolon.
Please Note: Only individuals with an active subscription will be able to access the full article. All other readers will be directed to the abstract and would need to subscribe.