Other Therapy Areas
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.
Aesthetics
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Revance Wins First Daxxify Therapeutic Indication, Plans Limited Initial Rollout
Similar to its post-approval strategy in aesthetics less than a year ago, Revance will initially target a small number of doctors as it Daxxify to market in the US for cervical dystonia with a broader launch in 2024.

BiologicsMD Uses Unique Fusion Proteins To Treat Alopecia Safely
Emerging Company Profile: The US biotech is developing a series of recombinant fusion proteins to tackle alopecia and other diseases without supressing the immune system.

Finance Watch: Brent Saunders Plots Return To Deal-Making With A SPAC IPO
Public Company Edition: The former Allergan CEO is taking a special purpose acquisition corporation public to generate $460m for deals in familiar biopharma niches. Also, IPOs are expected to keep up a robust pace in the fall and Albireo raises $160m on the success of its drug for pediatric liver diseases.

ITC Takes Medytox’s Hand In US Botulinum Case, Bans Daewoong’s Imports
USITC’s initial decision in the Medytox and Daewoong botulinum dispute is favorable to Medytox and poised to take a toll on the Korean firm and partner Evolus as they face a 10-year ban on imports of their product to the US.
Blood and Coagulation Disorders
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Pfizer Matches Beqvez Price To Hemgenix In Hemophilia B, Plans Warranty Program
The drug maker said the program would allow for continued reimbursement when patients change insurance plans. A Sangamo-partnered gene therapy for hemophilia A is also in the works.

Takeda Charges Into Diversification With ITP Win
The company announced positive topline results from a Phase II study of mezagitamab and expects to have five programs in Phase III development this fiscal year.

Things Get Worse For Gilead’s Magrolimab
Gilead said the FDA requested a partial clinical hold on the studies as well, just over a week after the drug maker said it would not develop the magrolimab further for blood cancers.

Grifols Aims For Largely Untapped Market With Successful Fibrinogen Deficiency Study
The Phase III trial of BT524 produced positive topline results in patients with acquired fibrinogen deficiency, potentially making it the first fibrinogen concentrate to win US FDA approval for that indication.
Dental Oral
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Start-up AFYX Applies Ultra-Sticky Patch Technology To Underserved Oral Mucosal Disease
Emerging Company Profile: Patients with oral lichen planus suffer from painful lesions in the mouth that are hard to treat with topical steroids, leaving room for development of a new delivery method for the established clobetasol.
Dermatology
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

AbbVie Claims Rinvoq Superiority To Dupixent In AD
In a head-to-head study of Rinvoq versus Dupixent in atopic dermatitis, the AbbVie drug showed superiority on a composite endpoint measuring skin clearance and itch resolution.

Incyte Hopes To Augment Immune/Inflammation Pipeline With Escient Deal
Incyte will pay $750m to acquire privately held Escient and its first-in-class oral antagonists of Mas-related G protein-coupled receptors X2 and X4.

BMS Has A Strong Pipeline, But Access Challenges Remain
Bristol Myers Squibb’s head of major markets, Monica Shaw, wants to improve patients’ access to lifesaving therapies. And the group has several new products ready to roll.

Oruka Aiming At AbbVie, UCB In Psoriatic Indications
The third Paragon spinout, going public in a reverse-merger with ARCA, announced a concurrent $275m financing to help it develop optimized biologics as potential standards of care in psoriasis and psoriatic arthritis.
Ear
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Fennec Finds Ex-US Commercial Partner For Pedmark In UK’s Norgine
Norgine will market Pedmark, indicated to reduce risk of chemotherapy-related hearing loss in pediatric cancer, in the EU and UK. It will seek additional approvals in Australia and New Zealand.

Acousia To Refresh Stagnant Hearing Loss Space With New Potassium Channel Approach
The German biotech plans to prevent chemotherapy-induced hearing loss and treat chronic hearing loss by targeting a potassium channel in the ear’s sensory hair cells as it tackles a widespread but side-lined area.
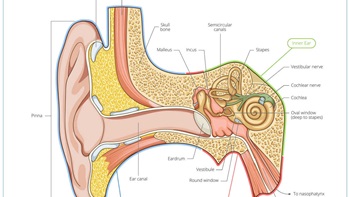
Lilly Builds On Gene Therapy Focus With Akouos Takeout
With the planned $487m acquisition of sensorineural-focused Akouos, Lilly makes its second gene therapy M&A play in two years, on top of partnering/financing activity in the space.

Otonomy’s Otividex Fails Again In Phase III Meniere’s Study
Otividex did not significantly reduce vertigo three months after treatment, so Otonomy appears to be moving on from Meniere’s disease and focusing on earlier-stage tinnitus and hearing loss candidates.
Ear Nose Throat
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.
Regeneron Strikes A CHORD With Deafness Gene Therapy
A very early success with a gene therapy for a rare form of deafness puts Regeneron ahead of Lilly and Sensorion.

Fennec Prepared For Market As Pedmark Finally Gets FDA Nod
After successive CRLs in 2020 and 2021, the drug gained a broad label for hearing loss in children treated with cisplatin for localized, non-metastatic solid tumors.

Optinose Looking For A Partner To Optimize Big Opportunity In Chronic Sinusitis
With positive Phase III data for its Xhance (fluticasone propionate) nasal spray in chronic sinusitis, Optinose believes it is poised to expand the target market for the drug 10-fold.

Budesonide CRL Another Setback For Takeda’s ‘Wave 1’ Pipeline
Setback for company's near-term launch goals and US patients with rare esophageal inflammatory condition as regulator asks for additional clinical data.
Gastrointestinal
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

MRM Health Pulls A Pouchitis Hit Out Of The Bag
The mid-stage success is encouraging, but taking on Takeda won’t be easy.

BMS’s Plans For Zeposia In Crohn’s Dubious Following Phase III Miss
The oral S1P modulator, approved to treat ulcerative colitis and MS, missed its primary endpoint in the first of two Phase III induction studies in moderate-to-severe Crohn’s disease.

Mirador Lands $400m To Pursue I&I Precision Medicine Approach
Ex-Prometheus team will use its big data platform to validate targets, select indications and optimize trial enrollment in the development of precision therapies for inflammatory and immune disorders.
.jpg?rev=67b89e260382428598594b7396006403&w=350&hash=47DBDB8D108A1854C56AD3F728FBD53D)
Ironwood Hits Phase III Endpoint In SBS-IF, But Shareholders Show Wariness
Looking to compete with Takeda’s Gattex in short bowel syndrome with intestinal failure, Ironwood succeeds in a Phase III trial, but investors may be concerned with misses on secondary endpoints.
Gynecology Urology
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Odd Coupling: J&J, RallyBio Collaborate As Only Firms Focused On FNAIT
While not co-developing each other’s drugs, J&J and financially troubled RallyBio are working on complementary approaches to the rare disease fetal and neonatal alloimmune thrombocytopenia.

Genmab Adds To Sector ADC Buying Spree With $1.8bn ProfoundBio Acquisition
The buyout reflects the Danish biotech’s plans to expand in oncology drug development and commercialization.

Scrip Asks…What Does 2024 Hold For Biopharma? Part 6: Therapeutic Area Advances
More than 100 biopharma executives and experts told Scrip their predictions for therapeutic area advances in the coming year. The recent commercial success of GLP-1s in diabetes and obesity and their potential in further disease areas fuelled excitement around the metabolic space. Expectations were also positive in neurology following the launch of Leqembi for Alzheimer’s disease in 2023, while the multiple opportunities to improve cancer treatment kept oncology top of the pile overall.

Freya Biosciences Has $38m And A Plan To Change Women’s Lives
Emerging Company Profile: Armed with a new tranche of funding, the Danish group is aiming to improve the lot of infertile couples by altering the microbiome of patients with dysbiosis.
Liver Hepatic
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

How Ochre Bio Bagged Boehringer For Its RNA-Based Regenerative MASH Therapies
UK-based Ochre Bio has signed its first major deal with Boehringer Ingelheim. Scrip talked to its co-founder and CEO, Jack O’Meara, about its human tissue-based drug discovery platform, its resulting RNA platform for liver disease and how the fledgling drug company's early work mirrored that seen in diagnostics.

Can Organovo Leverage Its Solid Phase II Data In MASH?
Cash-limited Organovo has an FXR agonist showing competitive efficacy and tolerability in MASH, but may need a partner to move forward in that disease as it may be prioritizing its IBD program.

A Good Choice To Have: Scrip’s Interview With Viking CEO Brian Lian
Still a clinical-stage company, Viking is studying promising candidates for both obesity and MASH in Phase II, but may need to prioritize one while partnering the other.
Madrigal Has A Slow, Steady Launch Planned For Rezdiffra
Madrigal faces the challenge of opening the metabolic-associated steatohepatitis (MASH, also known as NASH) market, but the firm has been laying groundwork for over a year.
Ophthalmic
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Falling Sales Force New Priorities At Roche
The Swiss major brings a new ruthlessness to the compounds in its pipeline: if they’re not first or best, they’re out.

Quick Listen: Scrip's Five Must-Know Things
In this week's podcast edition of Five Must-Know Things: Genmab buys into ADCs; Roivant set to take on AbbVie in uveitis; Ipsen signs firs ADC pact; South Korea looks to build biotech momentum; and the end of the road for Acorda.

LENZ Sees Big Commercial Market After Presbyopia Trial Success
LENZ announced successful results from its Phase III pivotal trial of LNZ100 and plans to apply for US FDA approval in the middle of this year, with commercialization in Q2 2025.

Roivant May Take On AbbVie In Uveitis With Strong Phase II Efficacy
Roivant’s brepocitinib bests data Humira previously posted in non-infectious uveitis, a top cause of blindness. The TYK2/JAK1 inhibitor also is in Phase III for dermatomyositis.
Orthopedics
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Ipsen’s Sohonos Clinches First US Approval For Ultra-Rare Bone Disorder
Just a month after an EU rejection, the French drugmaker’s oral retinoic acid receptor gamma agonist Sohonos has won a US thumbs up for the treatment of fibrodysplasia ossificans progressiva, an ultra-rare disorder that turns soft tissue into bone.

AstraZeneca’s Soliris Wins EU Recommendation In Pediatric gMG As Competition Looms
The UK major’s older C5 inhibitor product could soon receive EU approval for children and adolescents with the rare disease, but emerging competition from biosimilar developers and rival firms are starting to stack up.

Argenx Gets Under Skin Of Rivals With Vyvgart Hytrulo Approval
The US approval of a more convenient formulation of its myasthenia gravis therapy, Vyvgart, should cement its market position as well as change the definition of what being "well-controlled means for patients", Argenx hopes.

Inozyme Targets Underserved Mineralization Disorders Market With First-In-Class Enzyme Therapy
Emerging Company Profile: Fresh from a $73m follow-on offering last year, the US biotech is readying its lead asset for a pivotal trial in ultra-rare but debilitating mineralization disorders that affect infants and children.
Poisoning
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

SERB Pays $800m For Boston Scientific Specialty Pharma Business
The sale of BTG Specialty Pharmaceuticals to two affiliates of European specialty firm SERB leaves Boston Scientific with BTG’s interventional oncology and vascular products.

£80m To Tackle Snakebite Is A Wellcome Boost
Two major initiatives have been announced which hope to revolutionize the treatment of snakebites, which claims the lives of up to 138,000 people a year.

Indivior Pact Buys Addex Time For Progressing Dipraglurant And Pipeline
Addex Therapeutics says its licensing deal with Indivior to develop and commercialize its investigative addiction therapy ADX71441 'buys time' to find resources for progressing its own pipeline.
US Capitol Capsule: Move over Ebola! Fake drugs fight needs global attention, too
Much of the attention at the 2015 World Health Assembly (WHA) in Geneva, Switzerland later this month is expected to be focused on reforming the World Health Organization (WHO) and the failures by the international public health agency in responding to the ongoing Ebola epidemic in West Africa.
Renal
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Novartis Draws Out Its APPLAUSE
Another data drop from Fabhalta’s IgAN trial looks encouraging, despite a missing abstract.

Vertex’s $5bn Alpine Buyout Builds Autoimmune Disease Presence
The company hopes for a “Humira-like” blockbuster with povetacicept, the object of the sector’s biggest M&A deal so far this year.

Vertex Joins Regulus In Kidney Disease Therapy Hunt
The Boston, MA biotech is about to enter the clinic with its autosomal dominant polycystic kidney disease candidate but microRNA therapy pioneer Regulus is already setting the pace.
Novartis’s Fabhalta Hits In Glomerulopathy But How Hard?
So far, Novartis has only reported that its Phase III trial has succeeded and investors face a wait to find out just how good the drug is.
Wound Healing Tissue Repair
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Healios Acquires Bankrupt Athersys, Expands Cell Therapy R&D Programs
Tokyo-based Healios will lead the global clinical studies for Athersys’ cell therapy MultiStem for ARDS and start US Phase II trials for trauma following their long-term collaboration.

Swiss R&D Innovation: A Tale Of Two Firms Developing Cell Therapies For Wounds
While perhaps best known for historical achievements in immuno-oncology and CNS disorders, the Swiss biotech sector is slowly cultivating a new area of expertise in dermatology. Scrip spoke with two firms applying cell therapy technologies to the challenging area of chronic and acute wounds.

Vertex’s Type 1 Diabetes Cell Therapy Gets A Boost – And Some Competition
Vertex has invested heavily in advancing its stem-cell based candidates for type 1 diabetes, but Sernova has inched ahead in the race to bring a functional cure for the condition to patients.

Krystal Clears Path To Market For Topical Gene Therapy Vyjuvek
The company estimates there are about 9,000 reimbursable patients with dystrophic epidermolysis bullosa worldwide and anticipates a third-quarter launch following FDA approval.
You must sign in to use this functionality
Authentication.SignIn.HeadSignInHeader
Email Article
All set! This article has been sent to my@email.address.
All fields are required. For multiple recipients, separate email addresses with a semicolon.
Please Note: Only individuals with an active subscription will be able to access the full article. All other readers will be directed to the abstract and would need to subscribe.