
What Makes Asia Appealing For Clinical Trials?
Asia is continuing to emerge as a global hot spot for clinical trials, helped by positive factors including patient availability and competitive costs, but challenges remain, a recent conference in Seoul heard.

Green Cross Halts US, Europe GreenGene F Trials To Focus On China
Green Cross has overhauled the global development strategy for its hemophilia A therapy GreenGene F, halting slow-progressing Phase III trials in the US and Europe and shifting its focus instead to the fast-growing China market.
Celltrion Gets Phase IIb Green Light For Universal Influenza Antibody Drug
Hot on the heels of its biosimilar deal with Teva, Celltrion is also making a progress in development of novel drugs, receiving the green light for Phase IIb trial in South Korea for CT-P27, its in-house universal therapeutic antibody product for influenza.
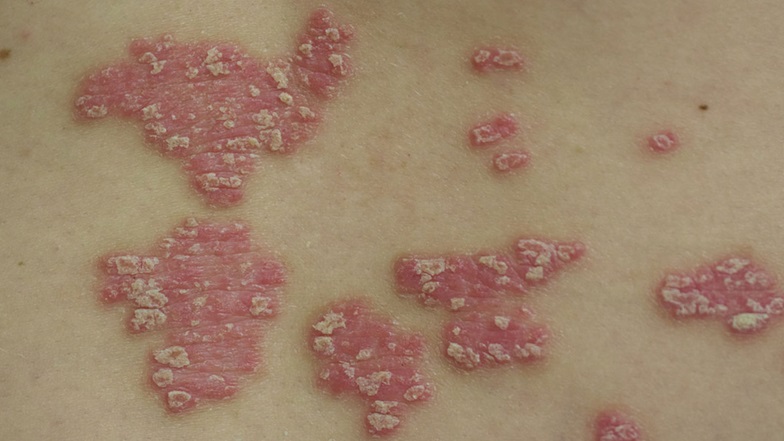
Sun’s Tildrakizumab Shines In Pivotal Psoriasis Trials
Sun Pharmaceutical, the world’s fifth-largest specialty generic company, has reported positive results from two pivotal Phase III clinical trials for its psoriasis candidate tildrakizumab, showing that the targeted biologic therapy delivers “significantly improved” skin clearance. While it will face rising competition, some analysts are predicting strong sales
Local Trials Could Delay India Dengvaxia Rollout By Three Years: Sanofi
In the face of a growing outbreak in the country, Sanofi has urged India’s government to waive its insistence on local Phase III clinical trials for its vaccine to prevent dengue, warning that insistence on the development program could delay the Indian introduction of Dengvaxia by three years, amid rising medical need.

Industry Sponsored Trials Fall Sharply In Challenging Indian Environment
A rise in bureaucracy and increased liability for clinical trial sponsors have impacted the popularity of India as a location for pharmaceutical companies’ clinical studies. Trialtrove’s Deborah Jeanfavre examines the data, which show a continuing decrease in new industry-sponsored trials starting in India over the past five years.

Indian Clinical Research Sector: Hope And Despair Amid Shutdowns
India's clinical research segment has had a rough time of late. Uncertainties, evolving regulations and compliance deviations have hurt the sector that is now scarred with firms downing the shutter, mistrust and constant speculation. Scrip spoke to a number of CROs who appear to have weathered the storm, but it seems the industry may not be out of the woods just yet.
How To Choose Clinical Sites In China (Part 2)
The situation for clinical trials at the execution level in China is very different from that in developed countries, given that sites are in hospitals and must be GCP-certified by the China FDA. Due to the limitations of study professionals, site management organizations have emerged as the solution to serve the rapidly increasing number of trials in the country following health care and regulatory reforms.
Oncology R&D Boosts Korean Clinical Trials In 2015
With South Korean pharma firms and drug developers striving to turn into innovators, the number of clinical trials approved by regulatory authorities climbed last year, led by studies for cancer therapies, particularly those acting on the immune system, one of the most active research areas globally.
New Orders Set To Clear Indian Trial Bottlenecks ‘Soon’
India's drugs regulator has indicated that bottlenecks impeding the flow of clinical trial-related activity in the country will be done away with, and that orders pertaining to a host of contentious clauses are likely soon.
How To Choose Clinical Sites In China (Part 1)
The situation for clinical trials at the execution level in China is very different from that in developed countries, given that sites are in hospitals and must be GCP-certified by the China FDA. Due to the limitations of study professionals, site management organizations have emerged as the solution to serve the rapidly increasing number of trials in the country following health care and regulatory reforms.
India Waives Large-Scale Trials For Sanofi’s Dengue Vaccine
An Indian panel has recommended the government dispense with Phase III trials for Sanofi's vaccine to prevent dengue after last year's major domestic outbreak of the virus caused by the same mosquito responsible for the current spread of Zika disease. The decision comes as India gears up to tackle the new threat from Zika.
You must sign in to use this functionality
Authentication.SignIn.HeadSignInHeader
Email Article
All set! This article has been sent to my@email.address.
All fields are required. For multiple recipients, separate email addresses with a semicolon.
Please Note: Only individuals with an active subscription will be able to access the full article. All other readers will be directed to the abstract and would need to subscribe.